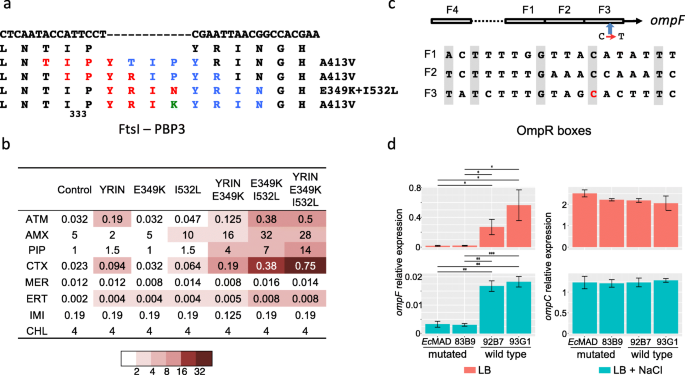
coli PBP3, with the exception that there was no influence on the MICs or minimal bactericidal concentrations of cefsulodin and cefepime, which have very low affinities for E. Similar results were obtained with PAO4089 overproducing E. aeruginosa PBP3 in PAO4089 did not affect susceptibility to PBP1-targeted cephaloridine or PBP2-targeted imipenem. aeruginosa PBP3 in PAO4089 resulted in twofold-increased resistance to aztreonam, fourfold-increased resistance to cefepime and cefsulodin, and eightfold-increased resistance to ceftazidime, whereas overproduction of the P. Susceptibility studies indicated that overproduction of the P. aeruginosa strain, PAO4089, that is deficient in producing chromosomal β-lactamase. These data are consistent with the conclusion that PBP3 has to achieve a certain level of saturation, with β-lactams targeted to this protein, to result in cell inhibition or death.Ībstract = "By using a broad-host-range vector, pUCP27, the Pseudomonas aeruginosa and Escherichia coli pbpB genes, which encode penicillin-binding protein 3 (PBP3), were separately overexpressed in a P.
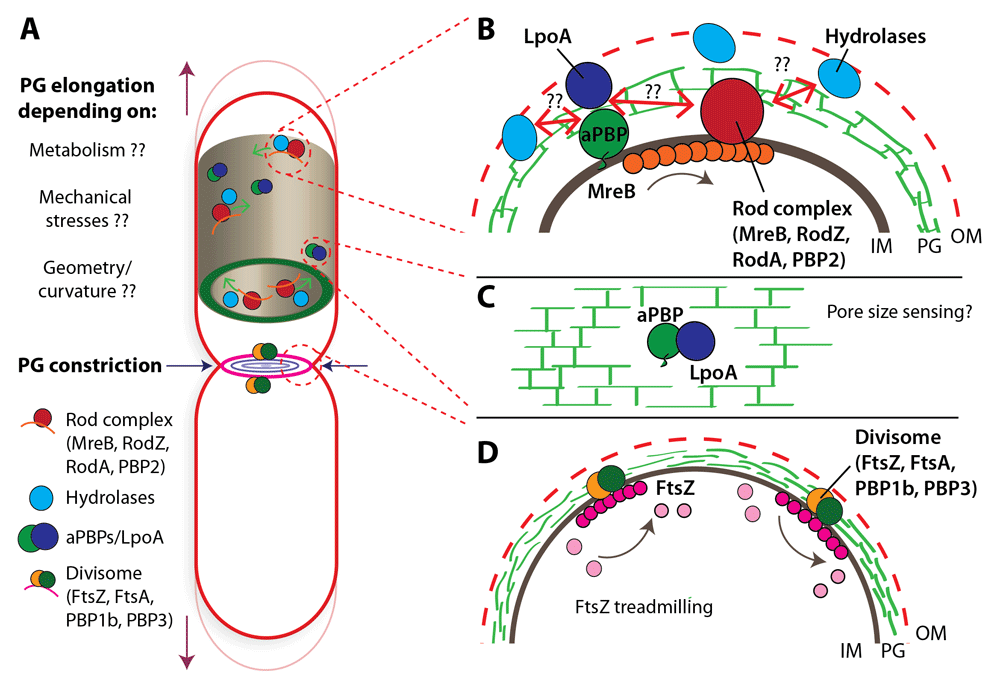

The structure shows a three dimensional domain swapping with a β-strand of one molecule inserted between two strands of the paired molecule, suggesting a possible role in PBP3(57-577) dimerization.Ĭentre d'Ingénierie des Protéines, Université de Liège, Institut de Physique B5a et Institut de Chimie B6a, Sart Tilman, Liège, Belgium.By using a broad-host-range vector, pUCP27, the Pseudomonas aeruginosa and Escherichia coli pbpB genes, which encode penicillin-binding protein 3 (PBP3), were separately overexpressed in a P. To gain additional insight, the PBP3 Val88-Ser165 subdomain (PBP3(88-165)), for which the electron density is poorly defined in the PBP3 crystal, was produced and its structure solved by SAD phasing at 2.1 Å. We have solved the crystal structure of a soluble form of PBP3 (PBP3(57-577)) at 2.5 Å revealing the two modules of high molecular weight class B PBPs, a carboxy terminal module exhibiting transpeptidase activity and an amino terminal module of unknown function. PBP3 is mainly periplasmic, with a 23 residues cytoplasmic tail and a single transmembrane helix. In Escherichia coli, penicillin-binding protein 3 (PBP3), also known as FtsI, is a central component of the divisome, catalyzing cross-linking of the cell wall peptidoglycan during cell division.
